Life Extension D-Ribose Tablets, plus calcium, helps maintain healthy energy levels in heart and muscle, encourages the production of ATP, gluten-free, non-GMO, vegan, 100 vegetarian tablets
US$9.60
Price when purchased online
Free shipping
Free 30-day returns
Sold and shipped by training.iaicb.com
We aim to show you accurate product information. Manufacturers, suppliers and others provide what you see here.
US$9.60
Price when purchased online
Free shipping
Free 30-day returns
Sold and shipped by training.iaicb.com
Free 30-day returns Details
Product details
| Management number | 220580409 | Release Date | 2026/05/03 | List Price | US$9.60 | Model Number | 220580409 | ||
|---|---|---|---|---|---|---|---|---|---|
| Category | |||||||||
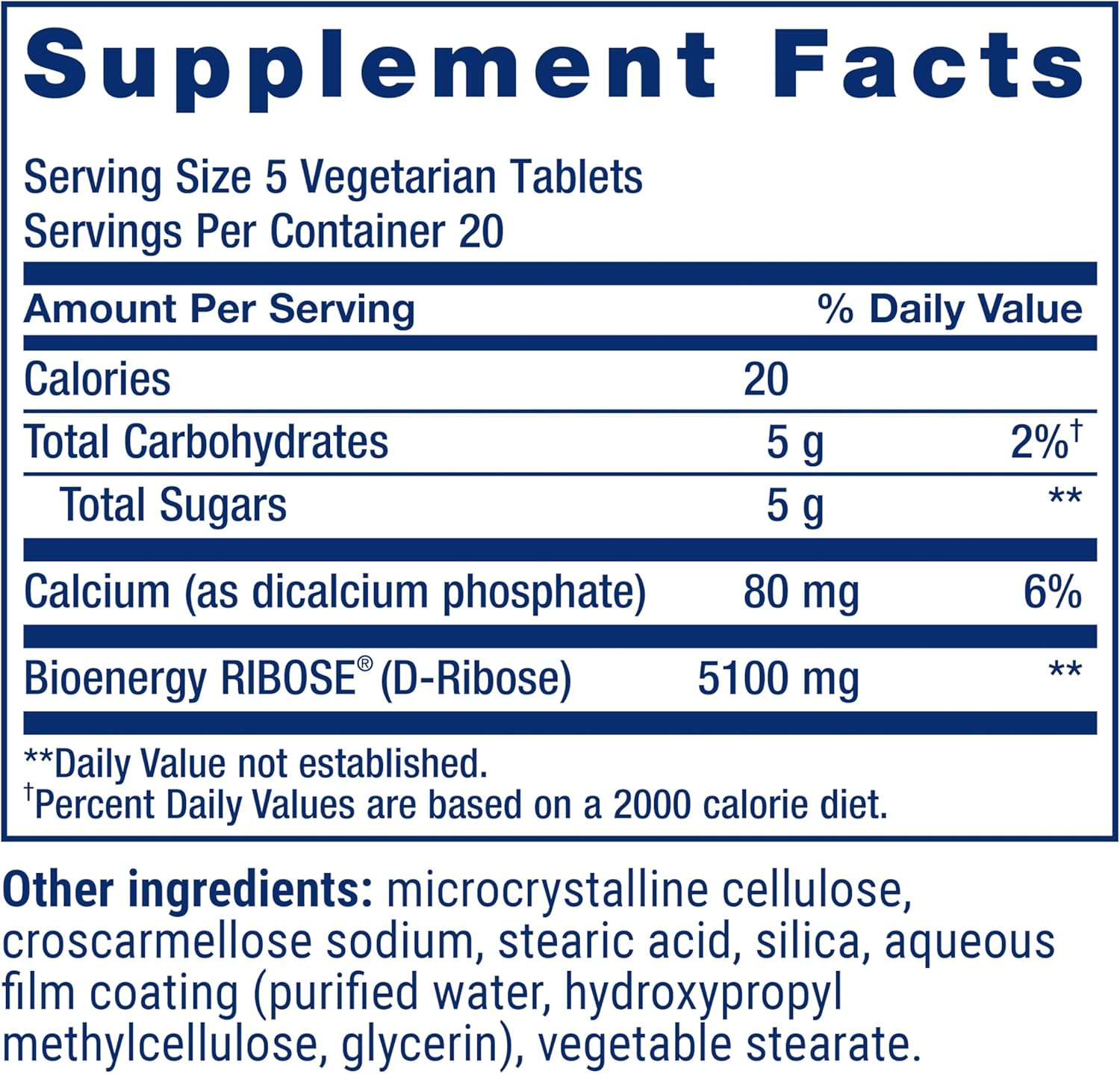
D-Ribose Tablets is an energy management formula. Each serving delivers 5100 mg of D-ribose plus 80 mg of calcium to encourage cellular energy production and support a healthy heart and muscle tissue. D-Ribose Tablets contains 100 vegetarian tablets, enough for a 20-day supply. It should be taken as five tablets daily with food to support cardiovascular health and energy levels in working muscles. Alternatively, take one tablet daily for general health. Continued use is necessary to maintain the benefits of D-ribose supplementation. This vegetarian and gluten-free formula is made with non-GMO ingredients.
- Encourage cellular energy production – D-Ribose Tablets help support your levels of ATP (adenosine triphosphate), which is critical for promoting cellular energy throughout your body.
- Post-workout energy boost – Looking to support energy levels in working muscles? D-ribose supplementation promotes exercise endurance and healthy cellular energy after exercise.
- For heart and muscle health – D-Ribose Tablets offer a convenient way to encourage ATP production, which supports both heart and muscle health.
- Ingredients matter – We use the finest raw ingredients with the quality and purity you deserve. The majority of our products are non-GMO, gluten-free and manufactured in the USA—and a Certificate of Analysis is available for every one.
- Live your healthiest life – We believe the answers to a healthier life are in reach, and that scientific research is the key to getting there. That’s why so many of our formulas feature specific ingredients and amounts based on scientific studies.



| UPC | 737870147336 |
|---|---|
| Color | Off White |
| Flavor | Unflavored |
| Diet Type | Gluten Free, Vegetarian |
| Item Form | Tablet |
| Brand Name | Life Extension |
| Model Name | D-Ribose Tablets |
| Unit Count | 100 Count |
| Dosage Form | Tablets |
| Item Weight | 0.43 Pounds |
| Manufacturer | Life Extension |
| Model Number | 01473 |
| Container Type | Bottle |
| Item Nype Name | D-Ribose Supplement |
| Item Dimensions | 2.5 x 2.5 x 5 inches |
| Number of Items | 1 |
| Product Benefits | Helps encourage cellular energy production, Helps replenish healthy cellular energy after exercise, Helps support healthy heart & muscle tissue |
| Material Features | GMO Free |
| Material Type Free | Gluten Free |
| Special Ingredients | Bioenergy RIBOSE® (Non-GMO D-Ribose), Calcium (as dicalcium phosphate) |
| Allergen Information | Gluten Free |
| Age Range Description | Adult |
| Primary Supplement Type | Calcium, D-Ribose |
| Manufacturer Part Number | 01473 |
| Specific Uses For Product | Active Lifestyle & Fitness, Energy Management, Heart Health |
| Total Servings Per Container | 20 |
| Global Trade Identification Number | 00737870147336 |
Correction of product information
If you notice any omissions or errors in the product information on this page, please use the correction request form below.
Correction Request Form